 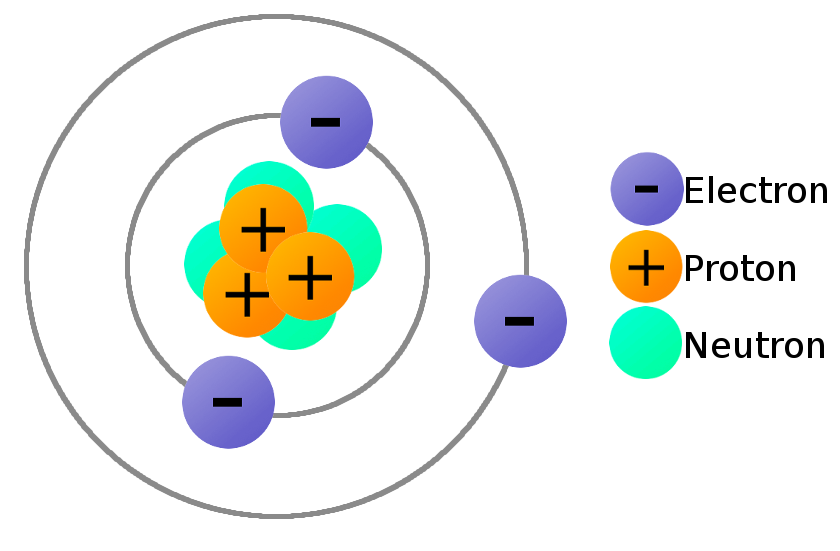
The term residual is associated with the fact, it is the residuum of the fundamental strong interaction between the quarks that make up the protons and neutrons. In nuclei, this force acts against the enormous repulsive electromagnetic force of the protons. Inside the atomic nucleus, the residual strong force, also known as the nuclear force, acts to hold neutrons and protons together in nuclei. Inside the protons and neutrons, we find the quarks. The nucleus itself is generally made of protons and neutrons but even these are composite objects. An atomic nucleus and an electron cloud, which are bound together by electrostatic force. In other words, the nucleus occupies only about 10 -12 of the total volume of the atom or less (the nuclear atom is largely empty space), but it contains all the positive charge and at least 99.95% of the total mass of the atom.Īfter discovery of the neutron in 1932 by the English physicist James Chadwick, models for a nucleus composed of protons and neutrons were quickly developed by Dmitri Ivanenko and Werner Heisenberg.Īll matter except dark matter is made of molecules, which are themselves made of atoms. In Rutherford’s atom, the diameter of its sphere (about 10 -10 m) of influence is determined by its electrons. These experiments were a landmark series of experiments by which scientists discovered that every atom contains a nucleus (whose diameter is of the order 10 -14m) where all of its positive charge and most of its mass are concentrated in a small region called an atomic nucleus. These experiments were performed between 19 by Hans Geiger and Ernest Marsden under the direction of Ernest Rutherford. 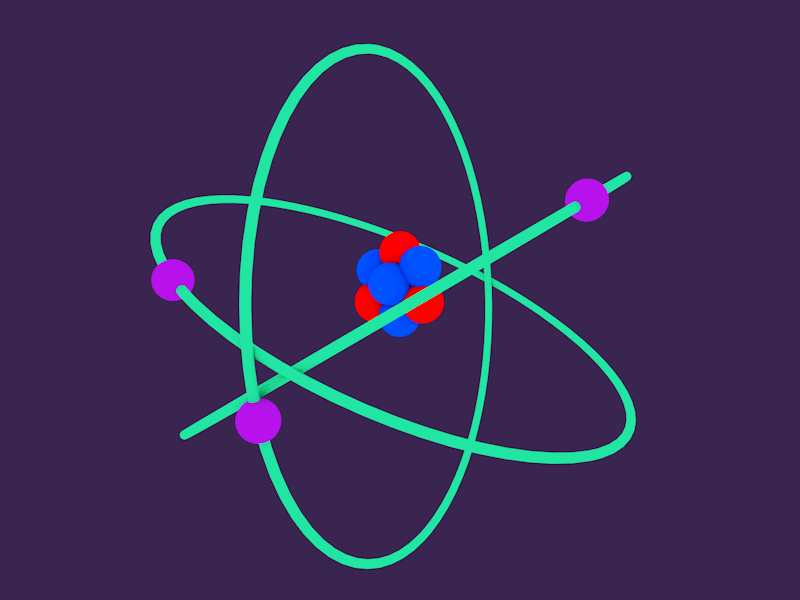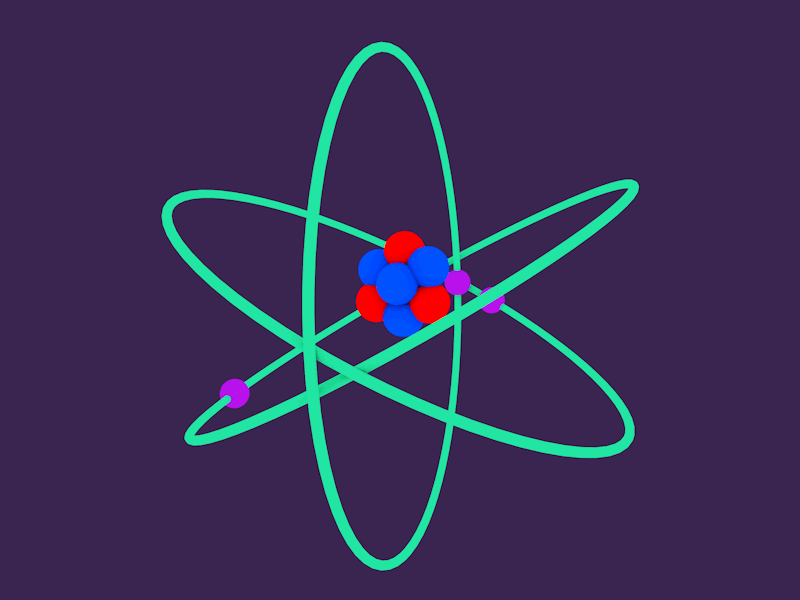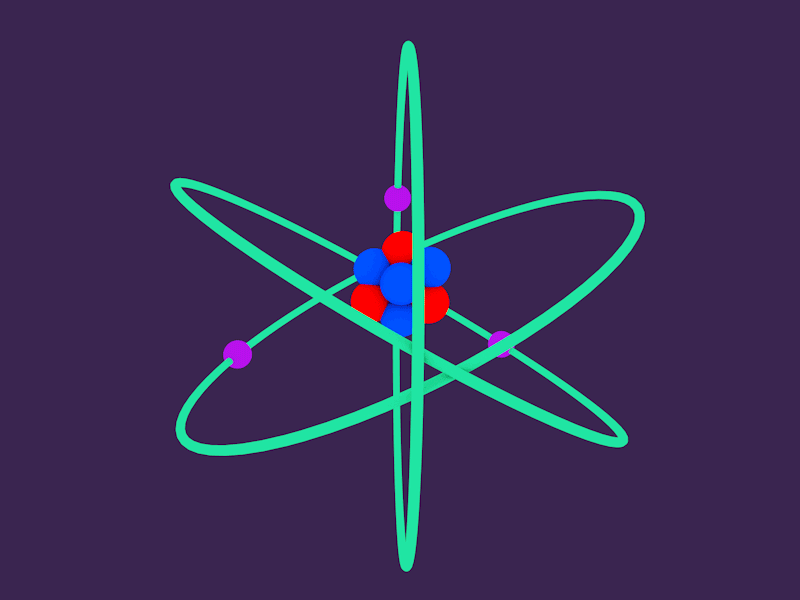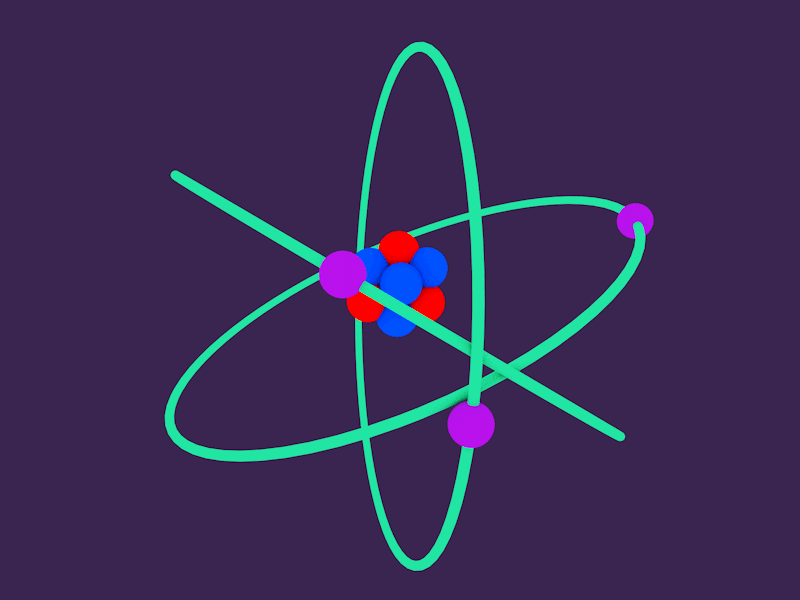
The atomic nucleus was discovered by Ernest Rutherford, who proposed a new model of the atom based on Geiger-Marsden experiments. The atomic nucleus also contains all of its positive electric charge (in protons), while all of its negative charge is distributed in the electron cloud. In comparison to an atom, it is much more smaller and contains most of the mass of the atom. In physics, the atomic nucleus is the central part of an atom. Protons and neutrons are most likely found in exactly the same space, at the central point. Join Seneca to get 250+ free exam board specfic A Level, GCSE, KS3 & KS2 online courses.Atomic Nucleus A figurative depiction of the helium-4 atom with the electron cloud in shades of gray. Want to learn more about Atom Size & Number? This information can be found for each element in the periodic table. The atomic number of an atom is the number of protons in the atom. The mass number of an atom (also called relative mass) is the total number of protons and neutrons in a nucleus. Relative mass of electrons = 0.0005 (this gets rounded to zero).Ītoms: Relative mass = number of protons + number of neutrons. 
**Mass: **Sub-atomic particles: Relative mass of protons and neutrons = 1. 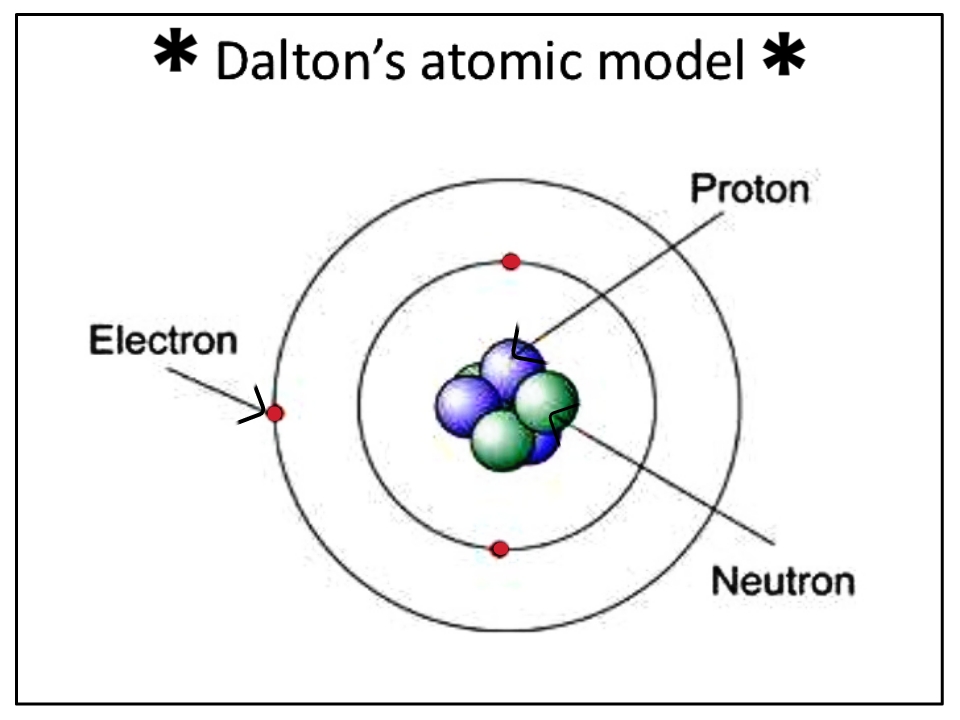
If an atom was the size of Wembley Stadium, then the nucleus would be the size of a garden pea. The nucleus of an atom is 10,000 times smaller than the atom. About 5 million hydrogen atoms could fit into a pin head. **Size: **Atoms have an average radius of about 0.1 nm. They are made up of sub-atomic particles (protons, neutrons, and electrons), which are even smaller and lighter than an atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
